
We have heard about hydrogen being a better, more eco-friendly power source for combustion engines than gasoline. Hydrogen combustion engines are similar to gasoline engines in many points.
It works exactly like the conventional gasoline combustion engine in the 4-stroke cycle. It could also use a direct or port fuel injection system. Read more about fuel injection systems here.
So they look very similar, so where do they differ? In the following lines, we will take a look at the 8 differences between a hydrogen combustion engine and a gasoline combustion engine.
1- Eco-friendly

Let begin with the most known point of hydrogen engines. Hydrogen is not a completely clean fuel for an engine, no matter how many claims to the opposite you may have heard.
In theory, the combustion process should be clean and eco-friendly and produce just water as a waste product. However, the high combustion temperatures will produce the pollutant nitrogen oxide (NOx), which is being eliminated by every automaker on the planet. The good news is that there are no CO2 emissions.
2- Better Air/Fuel Ratio

Hydrogen engines could operate on air to fuel ratio of up to 180:1, while gasoline engines can operate at a maximum 37:1 ratio. As a result of its leaner operating conditions, the hydrogen engine is much more easier to have a cold start since less fuel is needed.
This also means that hydrogen will need less fuel, resulting in significantly higher efficiency. Running that much lean will also reduce NOx emissions, which is one of the hydrogen engine drawbacks. However, going that much lean means generating less amount of power.
3- Low Ignition Energy
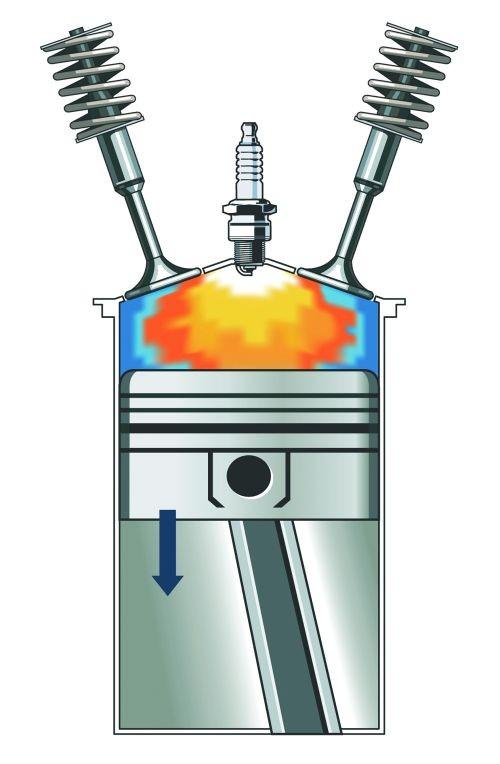
Ignition Energy is the amount of energy needed for a fuel mixture to ignite. The less energy needed, the easier it is to ignite.
Hydrogen, in general, needs a lot less than gasoline to ignite. It needs just about 0.02 mJ to ignite, while the gasoline would need 0.24 mJ.
This means it is easier to ignite that lean hydrogen-air mixture. But this has drawbacks on the engine performance. Usually, in the cylinders, we will have some hotspots that could lead to the ignition of the mixture earlier, which is known as misfiring.
4- Higher Ignition Speed
Hydrogen has a high ignition speed or what is known as flame velocity. This means that the flame will propagate faster within the cylinder, combusting the whole mixture faster.
This means that the engine will have higher efficiency at higher RPMs. But unfortunately, this advantage will be reduced as we go leaner in the mixture.
5- Higher Auto-Ignition Temperature

Auto-Ignition temperature is the temperature at which the air-fuel mixture will automatically combust at without the need for a spark. Hydrogen has a higher auto-ignition temperature of more than 500°C, but gasoline is just about 280°C.
And this means that we have the ability to compress the hydrogen mixture more than the gasoline mixture before it ignites. Having a higher compression ratio usually means more power and higher efficiency.
6- Better Mixing

Having a higher “diffusivity” means that the air and fuel mixture will be mixing better and faster. Hydrogen usually has a higher diffusivity rate than gasoline. This means that hydrogen engines work very well with a direct injection system since the fuel will quickly mix with the air in the combustion chamber.
This will usually lead to a more uniform mixture which means that we will have complete combustion leading to higher efficiency.
7- Lower Quenching Distance
Quenching distance is the distance between the cylinder walls and the maximum point the combustion flames reach. Hydrogen engines usually have a lower quenching distance, and this helps spread out the fuel to burn better, but it might also create a backfire because the hydrogen can fit between the valve and the combustion chamber more easily.
8- Lower Density

Compared to other fuels, hydrogen is highly dense in terms of energy to volume. That implies it occupies 30% of the combustion chamber, while the more power-dense gasoline will occupy just 1-2% of the area in a combustion chamber, which decreases the hydrogen engine power output.
To perform the same amount of work as gas, it requires more hydrogen. A hydrogen fuel tank also takes up a lot of room, which means that if you want to go the same distance as a gas-powered car, you may have to sacrifice some of your trunk and passenger space.
These are some of the most important differences between hydrogen and gasoline combustion engines. Do you think that we should switch to hydrogen as a fuel in our combustion engines? Please share with us your thoughts.
Jason Fenske from Engineering Explained youtube channel explained these differences in details, please take a look at his video for more information.




