Tiny Particles power chemical Reactions
A revolutionary material based on carbon nanotubes can create power by absorbing energy from its surroundings.
MIT engineers have developed a new approach of producing energy using microscopic carbon particles that may generate a current just by contact with the liquid in their area.
According to the researchers, the liquid, an organic solvent, sucks electrons out of the particles, providing a current that may be used to drive chemical processes or power micro- or nanoscale robots.
“This mechanism is new, and this method of generating energy is new,” says Michael Strano, Carbon P. Dubbs Professor of Chemical Engineering at MIT. “This technology is interesting because all that is required is the flow of a solvent through a layer of these particles. This permits you to conduct electrochemistry without the need of wires.”
In a new paper explaining this phenomenon, the researchers demonstrated that they could utilize this electric current to drive an organic chemical process known as alcohol oxidation – an essential organic chemical process in the chemical industry.
Strano is the paper’s senior author, published in Nature Communications today (June 7, 2021). Albert Tianxiang Liu, an MIT graduate student, and Yuichiro Kunai, a former MIT researcher, are the study’s primary authors. Graduate student Anton Cottrill, postdocs Amir Kaplan and Hyunah Kim, graduate student Ge Zhang, and recent MIT grads Rafid Mollah and Yannick Eatmon are among the other contributors.
Unique characteristics
Strano’s study on carbon nanotubes — hollow tubes consisting of a lattice of carbon atoms with unique electrical characteristics — led to the unexpected finding. Strano proved for the first time in 2010 that carbon nanotubes might produce “thermopower waves.” When a carbon nanotube is covered with a fuel coating, moving bursts of heat, known as thermopower waves, move up the tube and generate an electrical current.
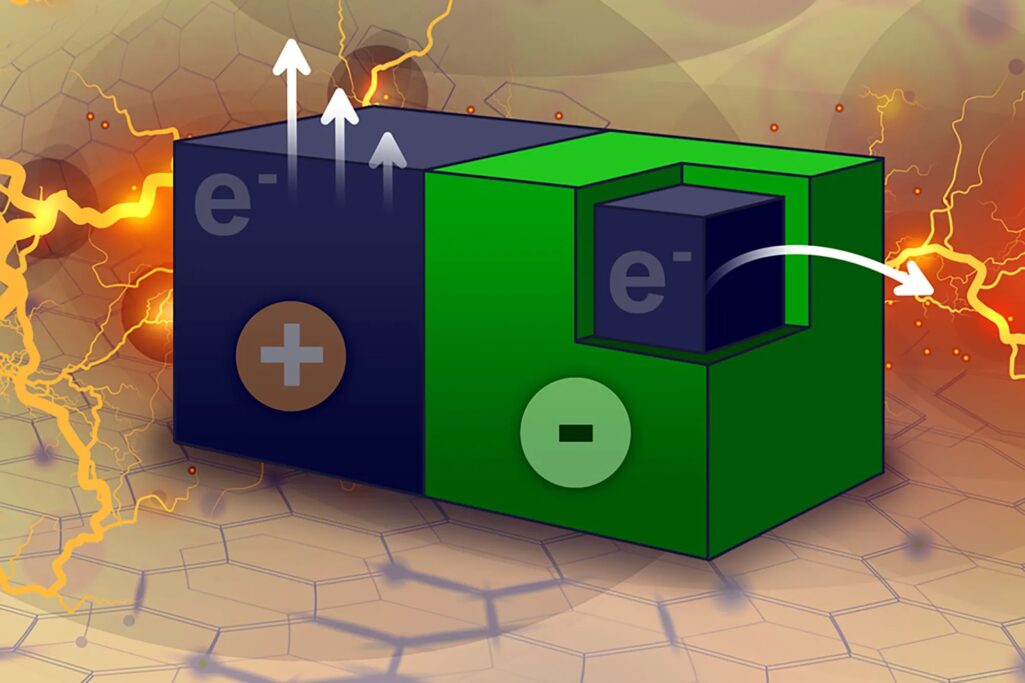
Strano and his students discovered a similar property of carbon nanotubes as a result of their research. They found that coating a portion of a nanotube with a Teflon-like polymer causes an imbalance that allows electrons to flow from the coated to the uncoated portion of the tube, resulting in an electrical current. These electrons can be extracted by immersing the particles in an electron-hungry solution.
To make use of this unique property, the researchers ground up carbon nanotubes and formed them into a sheet of paper-like material before shaping them into electricity-generating particles. The researchers coated one side of each sheet with a Teflon-like polymer before cutting off microscopic particles of any form or size. They created particles that were 250 microns by 250 microns for their study.
When these particles are immersed in an organic solvent, such as acetonitrile, the solvent sticks to the particles’ uncoated surface and begins to take electrons from them.
“The solvent removes electrons, and the system attempts to equilibrate by moving electrons,” Strano explains. “There is no advanced battery chemistry inside. It’s just a particle, and when you put it in a solvent, it begins to generate an electric field.”
“This study cleverly demonstrates how to extract the widespread (and often unnoticed) electric energy stored in an electronic material for on-site electrochemical synthesis,” states Jun Yao, an assistant professor of electrical and computer engineering at the University of Massachusetts at Amherst who was not involved in the study. “The beauty is that it points to a generic methodology that can be easily expanded to the use of various materials and applications in various synthetic systems.”
Particle energy
The particles in their current form may create around 0.7 volts of electricity per particle. The researchers showed that they could create arrays of hundreds of particles in a tiny test tube in this study. This “packed bed” reactor produces enough energy to fuel an alcohol oxidation process, converting alcohol to an aldehyde or a ketone. Typically, this reaction is not carried out via electrochemistry since it would need an excessive amount of external current.
“Because the packed bed reactor is compact, it has more application flexibility than a large electrochemical reactor,” Zhang explains. “The particles can be made very small, and the electrochemical reaction can be driven without the use of any external wires.”
Strano plans to employ this type of energy generation in the future to create polymers using only carbon dioxide as a starting ingredient. He has already made polymers that can replenish themselves using carbon dioxide as a building ingredient in a solar-powered process in a related effort. This piece is inspired by carbon fixation, a series of chemical events that plants employ to create sugars from carbon dioxide while utilizing solar energy.
In the long run, this method might be utilized to power micro- or nanoscale robotics. Strano’s group has already begun developing small-scale robots that might be used as diagnostic or environmental sensors in the future. He finds the notion of scavenging energy from the environment to power these kinds of robots fascinating.
“It means you don’t have to put energy storage onboard,” he explains. “What we like about this mechanism is that you can get energy from the environment, at least in part.”