
We all know that the water boils when heated up to 100°C or 212°F. But is it possible to just put water in a tall cup or tube and get that same boiling effect?
Yes, it is possible, and “The Action Lab” YouTube channel has shown that using a tube that was raised until the water started boiling on a height of about 35 feet. But the question that arises: why would water boil at normal ambient temperature by just placing it into a long tube? Let’s find out…
Water Vapor Pressure

One fascinating “piece of information” about water is that you can find it in all three phases on the Earth’s surface: solid (ice), liquid water, and gas. Water vapor is the gaseous state of water.
Molecular motion occurs even when a glass of water is left on a table. Molecules that have sufficient heat energy can separate and fly free. The water molecule is no longer a liquid but rather a gas—water vapor.
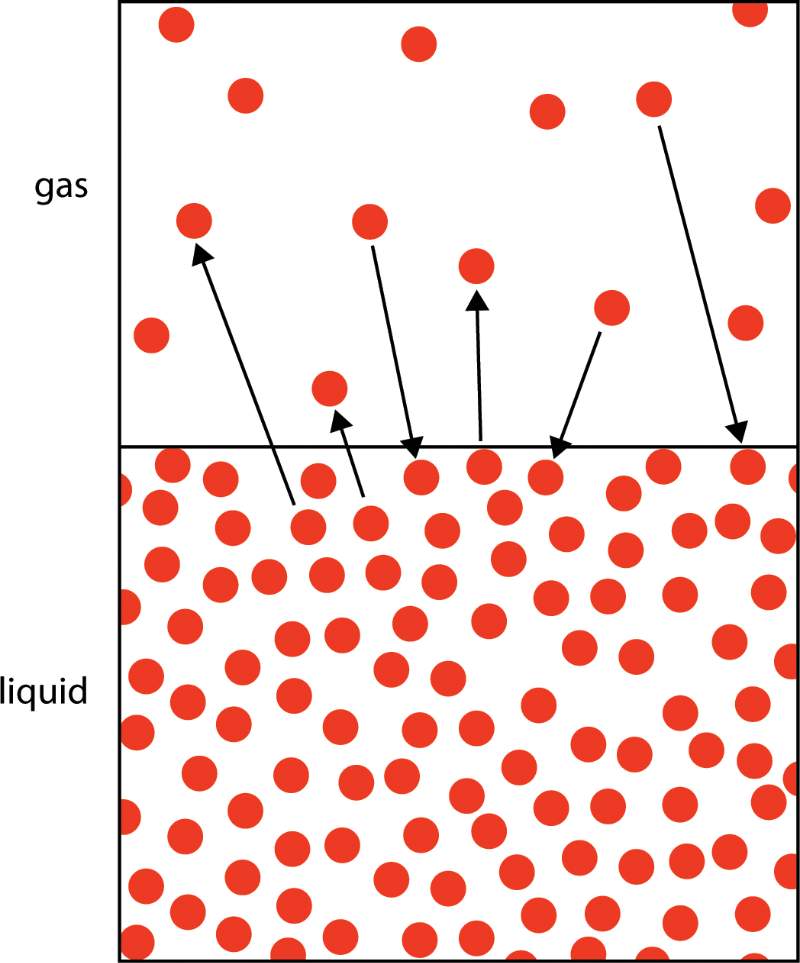
This water vapor may be obtained without the need for boiling. But hold on! It’s a two-way street. Gaseous water molecules can combine with liquid water to form new liquid water molecules. This is known as condensation.
In a sealed container, you may observe the condensation of water on the walls as a sign of this water vapor.

More and more water particles have enough energy to leave the water phase and form water vapor as the temperature increases.
Boiling Water

Now comes the boiling water. What does the water look like when heated to a temperature of 100 degrees Celsius? Bubbles everywhere!
Are the bubbles formed of air? A little hydrogen and oxygen wouldn’t hurt, either.
No. The bubbles are made of water vapor, which is tiny amounts of water in the gaseous state. They can’t be made of air, after all. Water vapor is the only logical explanation for the bubbles.
What, then, is causing these water vapor bubbles to pop? Temperature and vapor pressure are the only factors that matter. The average speed of water particles rises is directly proportional to the rise in water temperature.
To generate a burst of bubbles, water molecules in the liquid phase must have enough energy to push back against other water molecules. However, the water must be heated enough for the particles to move quickly enough.

But hold on! The vapor pressure is also a factor. There must be equivalent pressures inside and outside of the bubble in order for the bubble not to break apart. The vapor pressure is within the bubble, while the water pressure is on the other side. This implies that in order for water to boil, the temperature must rise until the vapor pressure equals the outside pressure and a bubble forms.
Decreasing the Pressure

The fun is about to begin. What would happen if I reduced the pressure exerted on some water in a liquid state? As a result, the liquid’s pressure would be reduced as well.
If lowered sufficiently, this pressure may be brought down to the same level as the vapor pressure. Boom. It’s no longer necessary to raise the temperature of the water in order for the bubbles to develop.
Even at room temperature, we can bring water to a boil. This is what The Action Lab did in his video. He first filled the tube entirely with water and then submerged one end in water gallon, and the other end was closed.
He then started lifting the tube through the levels of a parking structure. After reaching approximately 35 feet (10 m), the water started boiling. This is due to the decrease in pressure that happened in the water until reaching the water boiling point at the normal ambient temperature.
To see the experiment completely, watch the following video:




